A new study led by Souradeep Das and colleagues suggests that evolution primitive membranes may have been a key component of early evolution towards living systems. Published in Dynamical interactions among protocell populations: Implications for membrane-mediated chemical evolution in Philosophical Transactions of the Royal Society B, the research was conducted at the Indian Institute of Science Education and Research, at Pune. It presents experimental evidence that simple protocell membranes of distinct properties can compete, cooperate, and chemically evolve when placed together in the same environment.
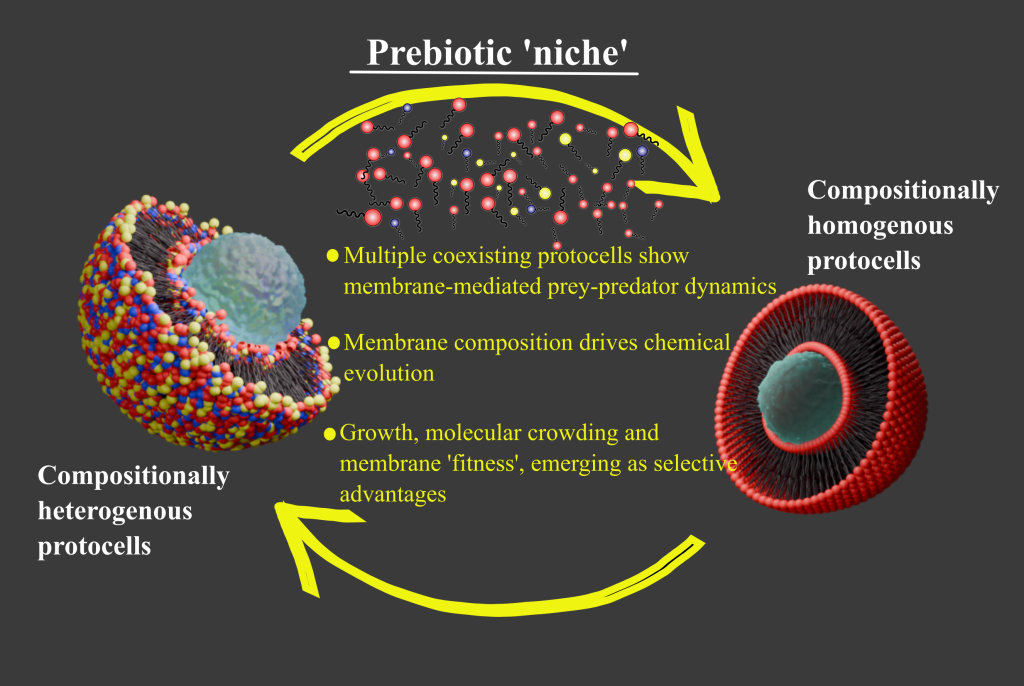
The findings challenge a long-standing assumption in prebiotic chemistry wherein most laboratory studies have focused on investigating a single kind of protocell (compositionally speaking). Das and colleagues instead recreated a more realistic representation of a chemically diverse prebiotic niche. The result was a striking predator-prey-like dynamic between different protocell populations, with consequences for chemical evolution, growth and molecular crowding.
Protocells before biology
In the context of origins-of-life research, protocells are model compartments formed from simple amphiphiles. On the early Earth, fatty acids and related derivatives could have spontaneously self-assembled in water to form membrane-bound vesicles. These primitive cell-like spheres can encapsulate solutes and concentrate biomolecules, creating microenvironments distinct from the surrounding bulk solution.
The membranes examined in this study were composed of single-chain amphiphiles, like oleic acid, oleyl alcohol, and glycerol monooleate. These molecules are widely considered prebiotically plausible. Under appropriate pH and buffer conditions, they self-assemble into lipid vesicles that mimic specific biophysical properties of biological membranes, albeit without proteins or genetic machinery.
Membranes are central to prebiotic Darwinian evolution because they impose boundary conditions. By separating inside from outside, they enable ‘selfness’, along with providing selective permeability, concentration gradients and localised reaction networks. Without compartments, the dilution problem severely limits the efficiency of prebiotic reactions. The question, however, is not simply how membranes formed but how they interacted in chemically heterogeneous environments.
A prebiotic niche with multiple membrane ‘species’
The study designed an experimental system that simulated a multispecies protocell niche. Four membrane populations were constructed. A homogenous population composed solely of oleic acid served as a control. Two binary populations contained oleic acid combined with either oleyl alcohol or glycerol monooleate. A tertiary population incorporated all three amphiphiles.
All vesicles were formed under identical conditions in bicine buffer at pH 8.5. Notably, there was no imposed environmental bias. Temperature, ionic strength, and pH were held constant. This allowed the researchers to isolate membrane composition as the key variable driving population-level behaviour.
When chemically distinct protocell populations were mixed, the results diverged dramatically from those observed in control monospecies populations, where multiple protocells present are chemically similar. Turbidity assays and dynamic light scattering revealed that the tertiary membrane population grew at the expense of the homogenous population. In contrast, when vesicles of identical composition were mixed, no significant change occurred.
Chemical predator and prey
The authors describe this phenomenon using a predator-prey analogy. The tertiary vesicles acted as predators, incorporating monomers released by the homogenous prey population. The prey population correspondingly shrank. Significantly, this interaction was not driven by biological agency but by physicochemical differences in membrane composition.
Microscopy provided striking visual confirmation. The tertiary vesicles not only increased in size but also underwent shape deformation. Spherical vesicles expanded into prolate and tubular structures, sometimes reaching dimensions more than ten times their original size. The extent of growth depended on the availability of the prey population.
This behaviour was absent in the monospecies scenario. Even when vesicles differed in size, concentration or lamellarity, if their membrane composition was identical, no net growth occurred. The emergent predator-prey dynamic was therefore rooted in compositional heterogeneity. In a prebiotic context, where diverse amphiphiles coexisted, such interactions may have been commonplace.
Membrane growth and kinetic signatures
To probe the mechanism underlying growth, the team employed Förster Resonance Energy Transfer kinetics in a stopped-flow fluorimetric setup. By embedding a FRET pair within the tertiary membranes, they quantified changes in membrane surface area as vesicles incorporated additional amphiphiles.
The kinetic traces revealed single-phase exponential growth with a rate constant of approximately 0.033 s−1. This was slower and mechanistically distinct from previously described vesicle-micelle growth systems. In the control monospecies mixtures, the FRET signal remained unchanged, confirming the absence of membrane expansion.
Parallel experiments monitored the shrinking prey population. This prey population displayed a two-phase exponential decrease in surface area, consistent with rapid initial monomer loss followed by a slower phase in a residual subpopulation. The reciprocity of growth and shrinkage strongly supported the hypothesis that monomer exchange between membrane populations was indeed the driving force.
Desorption rates and membrane fitness
In this system, fitness is defined by monomer retention. Single-chain amphiphile membranes exist in dynamic equilibrium between membrane-embedded and free monomer states. The rate at which amphiphiles desorb into the surrounding solution influences membrane stability and growth potential.
Using a pH-sensitive , the researchers quantified the desorption rates of the membrane monomers. The homogenous oleic acid vesicles exhibited the highest monomer escape rate, while the tertiary population showed a lower rate, indicating greater monomer retention. After growth in the heterospecies mixture, the resultant membrane population displayed a strikingly lower desorption rate.
This observation suggests a form of membrane-mediated chemical evolution. Through interaction, the membrane composition shifted toward a more ordered, stable state. Laurdan generalised polarisation measurements confirmed increased membrane order and packing density in the growing predator population. In effect, competition within the niche drove the emergence of ‘fitter’ membranes.
Molecular crowding in the prey population
The fate of the shrinking prey population proved equally intriguing. Shrinkage reduces the internal volume of vesicles. If encapsulated solutes remain trapped, their concentration increases. This phenomenon, known as molecular crowding, has profound implications for prebiotic chemistry.
By encapsulating calcein at partially self-quenching concentrations, the researchers demonstrated increased quenching in the prey vesicles during interaction. The data indicated a several-fold reduction in internal volume, implying substantial concentration of luminal contents.
Molecular crowding is known to enhance RNA catalysis, influence protein behaviour and alter reaction kinetics. In a prebiotic environment, crowding could have accelerated template-directed replication or other nonenzymatic reactions. Thus, even the shrinking prey population gained an emergent functional advantage. Rather than being completely eliminated, it coexisted while experiencing a chemically beneficial state.
Life’s origin was probably not a story of one extraordinary protocell doing everything right. It was rather more feasible for a messy and random ecosystem of many simple compartments sharing tasks and exchanging molecules. To truly understand life’s beginnings, we must explore such complex interacting populations that interacted, competed, cooperated and evolved together as a system.
— Souradeep Das
Increasing complexity in three population systems
The researchers extended the paradigm to a three-population system. When a binary membrane population was introduced alongside the tertiary predator and homogenous prey, both chemically ‘fitter’ populations grew simultaneously at the expense of the prey.
Live imaging confirmed concurrent expansion of multiple membrane types. Although division events were not observed within experimental timeframes, the formation of elongated tubular structures suggests that environmental fluctuations could eventually trigger fission. Such dynamics resonate with models of primitive cell proliferation in wall-less bacterial systems.
The introduction of additional membrane diversity increased competitive complexity. In a realistic early Earth setting, numerous amphiphilic species would have coexisted. Computational modelling will be required to explore the full parameter space of such interactions. Nevertheless, the experimental demonstration establishes a proof of principle for multispecies protocell ecology.
Reference
Das, S., Pal, R., & Rajamani, S. (2025). Dynamical interactions among protocell populations: Implications for membrane-mediated chemical evolution. Philosophical Transactions of the Royal Society B, 380, 20250104. https://doi.org/10.1098/rstb.2025.0104