Water pollution remains one of the most urgent environmental science challenges of the twenty-first century. Industrial chemicals, household detergents, and synthetic additives continue to enter rivers, lakes, and oceans despite advances in wastewater treatment technology. Among these pollutants, fluorescent brighteners have gained increasing attention because of their persistence, high solubility, and potential biological effects.
A recent study led by Eri Yoshida at Toyohashi University of Technology, Japan, published in the journal RSC Applied Polymers, proposes a promising new method for removing these contaminants. The research article titled “Effective and sustainable removal of a model fluorescent brightener using bicarbonate loaded polyelectrolytes in a closed loop CO₂ cycle” describes a recyclable polymer system capable of capturing fluorescent brighteners from water efficiently while enabling repeated regeneration of the material.
The findings contribute to ongoing discussions about sustainable chemistry, advanced wastewater treatment, and circular environmental technologies. They also demonstrate how carbon dioxide, often viewed only as a greenhouse gas, may play a constructive role in pollution mitigation.
Why fluorescent brighteners matter
Fluorescent brighteners are widely used chemical additives found in detergents, textiles, plastics, cosmetics, paper products and even some food packaging. Their role is cosmetic rather than functional. They absorb ultraviolet light and reemit visible blue light, giving materials a brighter or whiter appearance. While this improves product aesthetics, it also introduces persistent synthetic chemicals into aquatic ecosystems.
Scientific evidence increasingly suggests these compounds should be considered emerging contaminants. Studies cited in the research indicate possible endocrine disruption, gene mutation risks and dermatological effects such as eczema. Some fluorescent brighteners also exhibit strong binding affinity to proteins in the human body, raising concerns about long-term biological accumulation.
Environmental monitoring shows that these chemicals are difficult to remove through conventional wastewater treatment processes. Their high water solubility and chemical stability allow them to remain mobile in aquatic environments. This has implications for drinking water safety, ecosystem health and global pollution management strategies.
The chemistry behind the innovation
The research focuses on a fluorescent brightener known as disodium 4,4′ bis(2 sulfostyryl)biphenyl, often abbreviated as DSBP. This compound is frequently used as a benchmark contaminant in environmental studies because of its widespread industrial presence and chemical stability. Removing DSBP from water has historically been challenging due to its resistance to biodegradation and the potential formation of toxic byproducts during chemical decomposition.
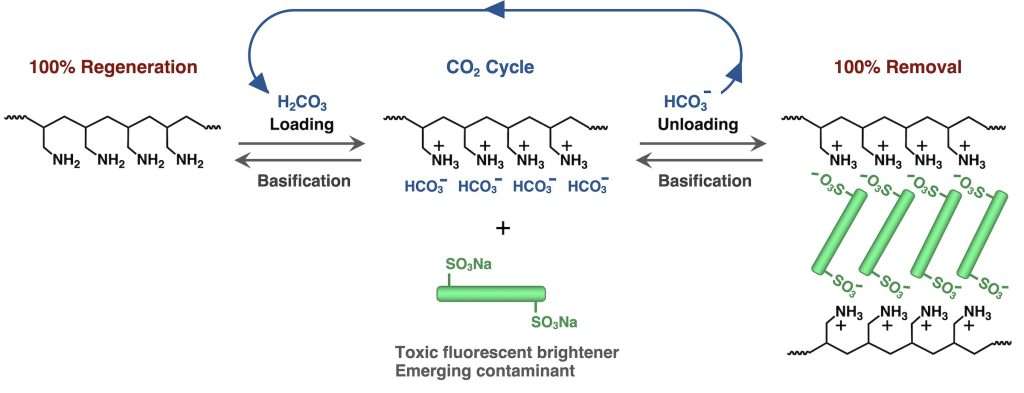
Yoshida’s study introduces a bicarbonate-loaded polyelectrolyte derived from poly(allylamine) (PAA). When carbon dioxide is introduced into an aqueous PAA solution, the polymer captures carbonic acid and forms poly(allylammonium bicarbonate). This transformation enables ion-exchange interactions between the positively charged polymer and the negatively charged sulfonate groups of fluorescent brighteners.
The interaction results in electrostatic cross-linking, effectively trapping the pollutant within polymer aggregates. According to the reported experimental data, removal efficiency approached nearly 100 percent regardless of polymer molecular weight. This level of efficiency is notable within the field of environmental polymer chemistry and suggests strong practical potential.
Closed-loop CO₂ cycling and regeneration
One of the most compelling aspects of the study is the reversible nature of the pollutant capture process. After the polymer captures DSBP, exposure to fundamental conditions causes the composite material to release the pollutant. This step regenerates the original polymer, which can then reload bicarbonate ions when carbon dioxide is reintroduced.n
This cyclical process establishes what the researcher describes as a closed-loop CO₂ cycle. From a sustainability perspective, this is significant because it reduces the need to continuously produce new adsorbent materials. Traditional adsorption technologies often struggle with regeneration efficiency, resulting in material waste and higher operational costs.
In contrast, the bicarbonate-loaded polyelectrolyte system demonstrated repeated capture-and-release cycles without significant loss of performance. The absence of toxic decomposition byproducts further strengthens its environmental appeal. These characteristics align with broader goals in green chemistry, resource efficiency, and climate-responsive engineering.
Broader environmental implications
While the study focused specifically on a model fluorescent brightener, the authors suggest that the approach could extend to other anionic pollutants. This includes sulfonated dyes, industrial surfactants, per and polyfluoroalkyl sulfonates and related chemical contaminants frequently detected in wastewater streams.
If scalable, the technology could support improved wastewater treatment in sectors such as textile manufacturing, food processing, cosmetics production, and medical materials manufacturing. These industries contribute substantially to chemical discharge into water systems. Effective removal technologies, therefore, hold both environmental and economic significance.
The integration of carbon dioxide utilisation into pollution control also adds an interesting dimension. Rather than treating CO₂ solely as an emission problem, this research frames it as a chemical resource that can facilitate environmental remediation. Such approaches are increasingly explored in carbon capture utilisation and storage initiatives worldwide.
Simply capturing CO₂ with absorbents can generate additional waste in the form of unrecyclable CO₂-laden materials. We need to shift toward developing new technologies driven by reversible CO₂ sorption that enable the efficient use of CO₂.
– Eri Yoshida
Technical validation and analytical insights
The study employed several analytical techniques to confirm pollutant capture and polymer behaviour. Ultraviolet-visible spectroscopy showed the disappearance of characteristic DSBP absorption bands after treatment, indicating effective incorporation into polymer composites. Light-scattering analysis revealed aggregation behaviour that depended on polymer chain length and pollutant concentration.
Thermogravimetric analysis and differential scanning calorimetry provided information about thermal stability and structural organisation of the composites. These findings suggested strong electrostatic interactions between the polymer matrix and the captured fluorescent brightener molecules. Electron microscopy images showed morphological changes consistent with the formation of a cross-linked composite.
Such detailed characterisation reinforces the robustness of the findings. It also demonstrates that the technology is grounded in established principles of polymer science, physical chemistry and environmental materials engineering rather than speculative innovation.
Remaining challenges before real-world adoption
Despite encouraging results, several practical considerations remain before widespread industrial implementation. Laboratory-scale efficiency does not always translate directly into large-scale wastewater treatment systems. Factors such as cost of polymer synthesis, energy requirements for pH adjustment and process integration must be evaluated carefully.
Another consideration involves regulatory approval and environmental safety assessments. Although the polymer itself is commercially derived, its modified form and regeneration cycle would require evaluation to ensure no secondary environmental impacts arise. Long-term durability and operational stability under variable wastewater conditions also require investigation.
Nevertheless, the research represents a meaningful step forward in sustainable water purification technology. Continued interdisciplinary collaboration between chemists, environmental engineers, and industry stakeholders will be essential to realise practical deployment.
A step forward for sustainable chemistry
This work contributes to a growing body of research that emphasises circular chemistry principles. Designing materials that capture pollutants while remaining recyclable addresses both environmental contamination and resource efficiency challenges simultaneously. It also reflects evolving priorities in environmental science research where sustainability, scalability, and lifecycle impacts are considered alongside performance metrics.
The study further illustrates how fundamental polymer chemistry can intersect with pressing environmental issues. By combining carbon dioxide utilisation, ion-exchange chemistry, and advanced material design, the researcher demonstrates a multidisciplinary approach increasingly necessary for tackling global pollution problems.
As awareness of emerging contaminants continues to grow, innovative technologies like bicarbonate-loaded polyelectrolytes may become essential components of future wastewater treatment strategies. While further validation is needed, the research provides a scientifically credible pathway towards cleaner water systems and more sustainable chemical management.
Reference
Yoshida, E. (2025). Effective and sustainable removal of a model fluorescent brightener using bicarbonate loaded polyelectrolytes in a closed loop CO₂ cycle. RSC Applied Polymers. https://doi.org/10.1039/d5lp00281h







