As the world accelerates towards electrification, the demand for efficient energy storage technologies has reached unprecedented levels. From portable electronics to electric vehicles and renewable energy systems, the need for devices that can store and deliver energy rapidly is more pressing than ever. While batteries dominate the conversation due to their high energy density, supercapacitors have emerged as a complementary technology owing to their rapid charging and discharging capabilities, long cycle life, and high power density.
However, supercapacitors are not without limitations. Their relatively lower energy density compared to batteries has restricted their widespread application in high-energy-demand scenarios. Researchers worldwide are therefore exploring innovative strategies to enhance performance without compromising their inherent advantages. A recent study presents a compelling advancement in this direction by introducing a magnetic-field-assisted approach to enhance electrochemical energy storage.
Published in RSC Applied Interfaces, the study titled “Magnetic-field-induced enhanced electrochemical energy storage performance of nickel cobalt phosphide” demonstrates how external magnetic fields can significantly boost the performance of advanced electrode materials. The research was conducted under the supervision of Prof. Manoj Kumar Singh, Central University of Haryana (CUH), Gov. of India, and Prof. Chandra Sekhar Rout, CNMS, Jain (Deemed-to-be University), Bangalore, India.
Understanding supercapacitors and their limitations
Supercapacitors, also referred to as electrochemical capacitors, occupy an important niche between traditional capacitors and batteries. They are capable of delivering high power density and exhibit excellent cycle stability, making them suitable for applications requiring rapid energy delivery. Their working principles involve either electrostatic charge storage, as in electric double-layer capacitors, or faradaic redox reactions, characteristic of pseudocapacitors.
Despite these advantages, achieving a balance between high energy density and long-term stability remains a major challenge. The performance of supercapacitors is largely determined by the properties of the electrode materials, including electrical conductivity, surface area, and ion transport dynamics. Transition metal phosphides have attracted attention in recent years due to their favourable electrochemical properties, including high electrical conductivity and rich redox chemistry.
Among these materials, nickel-cobalt phosphide, often abbreviated NiCoP, stands out for its bimetallic composition. The synergistic interaction between nickel and cobalt enhances electron transport and facilitates multiple oxidation states, thereby improving charge storage capacity. Yet, issues such as limited cyclic stability and slow ion diffusion continue to hinder its practical deployment.
A novel magnetic field approach
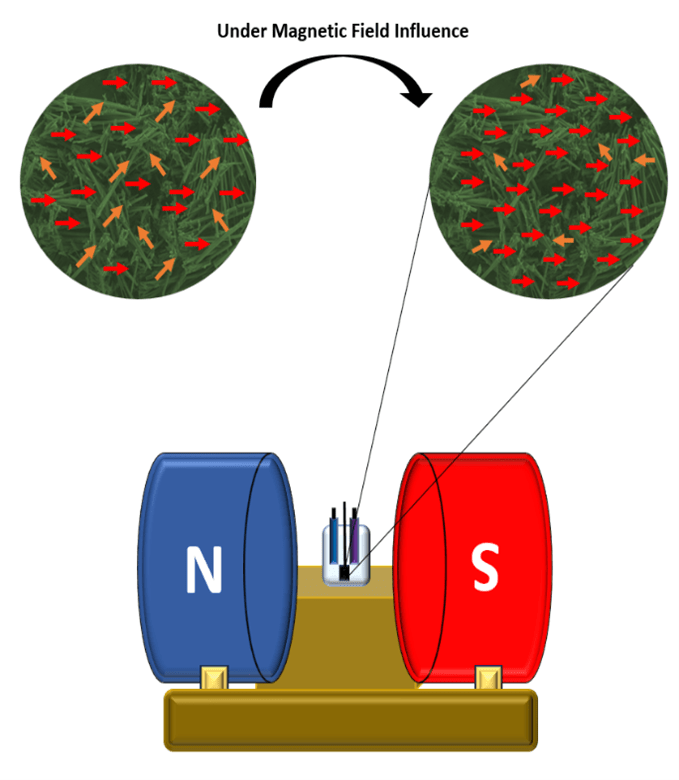
The study by Yadav introduces an innovative method to address these challenges by applying an external magnetic field during electrochemical operation. The underlying concept is rooted in the Lorentz force, which acts on charged particles moving in a magnetic field. This force can influence ion trajectories, enhance ion mobility, and improve the overall kinetics of electrochemical reactions.
To investigate this effect, the researchers synthesised NiCoP nanorods using a hydrothermal method followed by phosphorisation. These nanostructures were deposited onto a carbon paper substrate to form the supercapacitor’s working electrode. The electrochemical performance was then evaluated in an alkaline electrolyte under varying magnetic field strengths, as shown in Figure 1.
The results revealed a remarkable performance enhancement when a magnetic field was applied. At a field strength of 400 G, the specific capacitance increased from approximately 102 F/g to 264.528 F/g, representing an improvement of 160 percent. This significant increase underscores the potential of magnetic-field-assisted electrochemical systems to advance next-generation energy storage technologies.
Mechanisms behind the enhancement
The performance improvement can be attributed to several interrelated mechanisms. The application of a magnetic field reduces the thickness of the Nernst diffusion layer, thereby facilitating faster ion transport between the electrolyte and the electrode surface. This reduction enhances the accessibility of active sites and accelerates redox reactions within the electrode material.
Furthermore, the Lorentz force aligns magnetic dipoles within the material, thereby improving charge-transfer kinetics. The study also reports a substantial decrease in charge-transfer resistance, from 8.727 ohms in the absence of a magnetic field to 2.249 ohms at 400 G. This reduction indicates enhanced electrical conductivity and more efficient electron-transport pathways.
Another critical factor is the increase in ion diffusion rates. Under the influence of the magnetic field, electrolyte ions exhibit increased mobility, enabling deeper penetration into the electrode structure. This results in enhanced faradaic reactions and higher charge storage capacity. Collectively, these effects contribute to the observed improvement in supercapacitor performance.
Performance metrics and practical implications
In addition to specific capacitance, the researchers evaluated other key performance indicators, including energy and power density. The NiCoP-based supercapacitor achieved a peak energy density of 72.01 W.h/kg and a maximum power density of 4219.92 W.h/kg under a magnetic field. These values highlight the material’s potential for high-performance energy storage applications.
The device also demonstrated 100 percent coulombic efficiency, indicating excellent reversibility of the charge-discharge process. However, long-term stability tests revealed a decline in capacitance retention to approximately 57 percent after 5000 cycles under a magnetic field. While this suggests some degree of material degradation over time, it does not significantly affect the efficiency of individual charge-discharge cycles.
These findings suggest that while magnetic field-assisted systems offer substantial performance gains, further optimisation is required to enhance long-term durability. Nevertheless, the ability to achieve such high performance through a relatively simple external intervention represents a promising avenue for future research.
The external magnetic field offers a promising pathway to enhance supercapacitor performance by accelerating ion transport and improving charge storage efficiency.
—Manoj Kumar Singh
Insights from theoretical modelling
To complement the experimental findings, the study employed density functional theory calculations to investigate the electronic and magnetic properties of the NiCoP system. These simulations provided valuable insights into the role of spin configurations and magnetic ordering in influencing quantum capacitance.
The results indicated that a ferromagnetic configuration, corresponding to a strong magnetic field, exhibited higher quantum capacitance at positive voltages compared to non-collinear configurations. This behaviour aligns with experimental observations, reinforcing the conclusion that magnetic-field-induced spin alignment enhances electrochemical performance.
The theoretical analysis also highlighted the importance of the electronic density of states near the Fermi level, which plays a crucial role in determining charge storage capability. By correlating magnetic properties with electrochemical behaviour, the study offers a comprehensive understanding of the mechanisms driving performance enhancement.
The concept of using magnetic fields to modulate electrochemical processes opens new possibilities for designing advanced energy storage devices. This approach could be applied to a wide range of electrode materials, potentially leading to significant improvements in performance across various technologies.
Reference
Yadav, A., Baasanjav, E., Sahoo, M., Pradhan, K., Jeong, S. M., Singh, M. K., & Rout, C. S. (2026). Magnetic-field-induced enhanced electrochemical energy storage performance of nickel cobalt phosphide. RSC Applied Interfaces, 3, 431–441. https://doi.org/10.1039/d5lf00288e
Coauthors

Prof Manoj Kumar Singh is a Professor of Physics at the Central University of Haryana (CUH), Gov. of India. He completed his Master’s from Lucknow University and earned a PhD from IIT Bombay in Dec., 2004. He worked at the University of South Florida, USA for about one and a half years and received two US patents. Later, he served at the University of Aveiro (EU-Portugal) (2006 – 2018) as senior scientist. He has published over 150 international papers, with an h-index of 53 and 13,000 citations. He also won the FCT Starting Grant (2013), contributed to international conferences, and collaborated with global institutions.

Prof Chandra Sekhar Rout is a distinguished full professor at the Centre for Nano and Material Sciences (CNMS), Jain University. Prof. Rout earned his B.Sc. (2001) and M.Sc. (2003) degrees from Utkal University and completed his Ph.D. at JNCASR, Bangalore, in 2008 under the mentorship of Prof. C.N.R. Rao, Bharat Ratna awardee. He pursued postdoctoral research at the National University of Singapore (2008–2009), Purdue University, USA (2010–2012), and UNIST, South Korea (2012–2013). Prof. Rout serves as an associate editor for RSC Advances (Royal Society of Chemistry) and The American Journal of Engineering and Applied Sciences (Science Publications) and is a board member of various reputed journals.







